Northern Ireland Probes Two Deaths Linked to Weight-Loss Injections
Note: AI technology was used to generate this article's audio.
- Two possible deaths linked to weight-loss injections reported in Northern Ireland
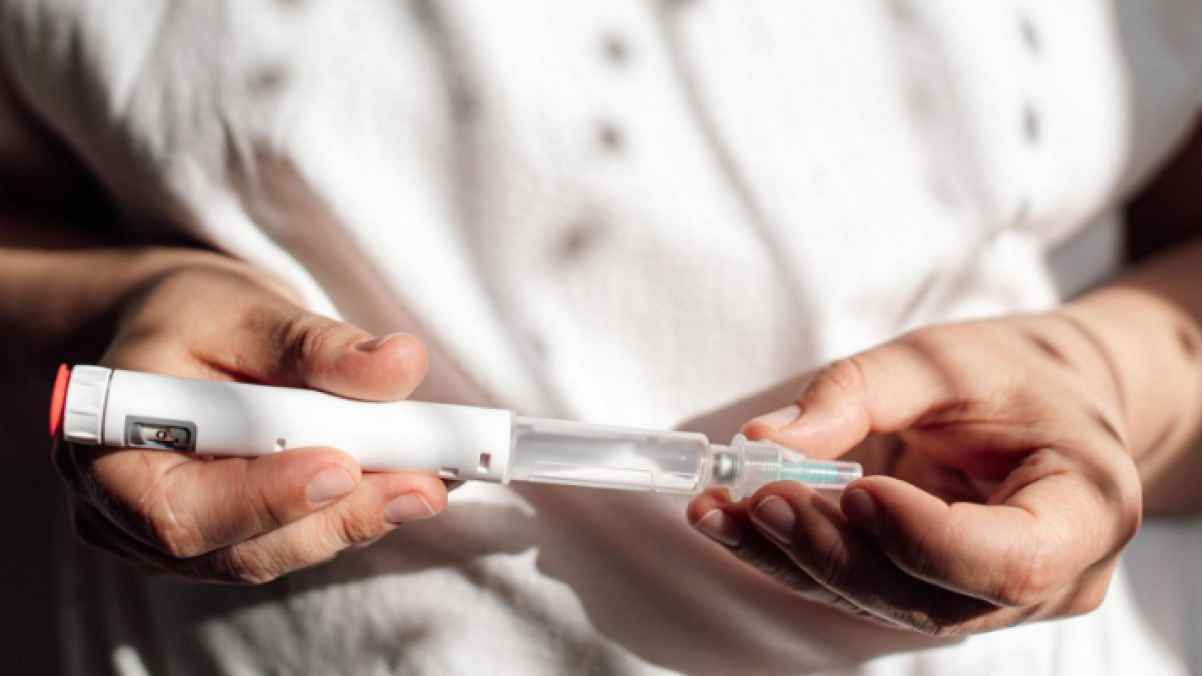
Health authorities in Northern Ireland have recorded two deaths that may be linked to weight-loss injections, according to a report submitted to the government’s medicines safety body.
These cases are part of more than 500 suspected side-effect reports related to medications submitted from Northern Ireland over the past two years, mainly involving GLP-1 drugs widely used to manage weight and treat diabetes in the UK.
The MHRA (Medicines and Healthcare products Regulatory Agency) emphasized that a suspected adverse reaction report does not necessarily mean the medicine caused the issue, only that the reporter suspected a link. Other underlying health conditions or coincidence could also explain the events.
The two cases involve a man and a woman, one in their 40s and the other in their 60s. About 82% of all suspected reports were submitted by doctors. One of the deaths was associated with the branded drug tirzepatide, while the other involved an unspecified semaglutide product.
The data also show six cases of acute pancreatitis over the past two years, a known side effect of GLP-1 drugs often linked to gallstones, as the MHRA warned in January.
According to a recent study by University College London, approximately 1.6 million adults in England, Scotland, and Wales used GLP-1 medications for weight loss between early 2024 and early 2025.
